Department of Health and Human Services | Food and Drug Administration
MEDWATCH
The FDA Safety Information and Adverse Event Reporting System
Form 3500-MAHA
Form Approved: Not Applicable
Section A
Patient Information
Section B
Adverse Event or Product Problem
Childhood vaccine schedule reduced from 17 to 11 recommended vaccines.
CDC Advisory Committee on Immunization Practices (ACIP) fired and reconstituted with Kennedy's picks.
Kennedy endorses Trump executive order boosting glyphosate production, after years calling it a carcinogen.
Federal judge rules vaccine schedule changes "probably illegal" and Kennedy "likely broke the law."
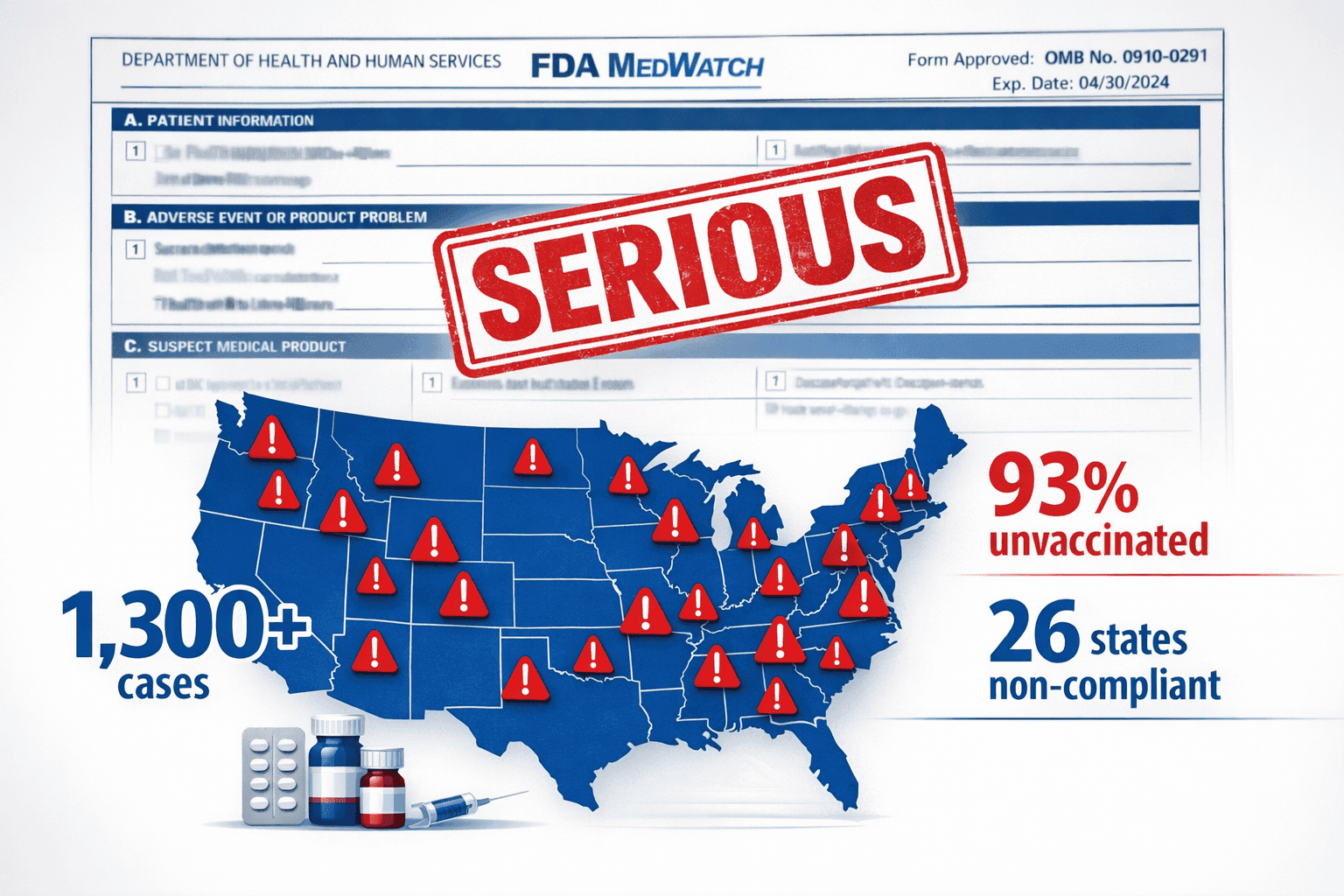
1,300+ measles cases in 2025 (93% in unvaccinated individuals).
The Lancet editorial: "The destruction that Kennedy has wrought in one year might take generations to repair."
Robert Malone angrily quits CDC panel, cites "weaponized leaking, sabotage."
Acting CDC Director Bhattacharya publicly supports measles vaccine, breaking from Kennedy.
26 states reject CDC vaccine guidance.
Section C
Suspect Product(s)
[Ref: Doc 01 - Methylene Blue]
Section D
Suspect Medical Device
Not applicable. Patient implementation of national policy objectives occurs without reliance on approved medical hardware.
Section E
Reporter Information
All reporters independently confirmed adverse events.
Outcome Assessment
Outcomes Attributed to Adverse Event
Loss of measles elimination status pending PAHO review. DTaP coverage dropped from 95% to 92.1%. 42% of Americans cannot name who runs the FDA.
Relevant Tests / Laboratory Data
Including Dates
Public Trust Polling
42%
of Americans cannot name the FDA commissioner.
Politico, March 2025
DTaP Coverage
95.0%92.1%
Declined from 95% (2019-20) to 92.1% (2024-25). Below herd immunity threshold.
Measles Incidence
1,300+
cases in 2025, on top of 2,200+ in 2024. Highest in three decades.
State Compliance
26
states rejected CDC vaccine guidance in the past year.
Personnel Status: Casey Means
Surgeon General Nomination
Confirmation stalled in Senate.
PendingPersonnel Status: Vinay Prasad
FDA Role
Leaving FDA end of April after “tumultuous tenure.”
DepartingThis form has been filed by the available evidence. No response is expected. No response has been forthcoming.
This page is satire formatted as a pharmaceutical document. It is not medical advice. The pharmacology, however, is real.